PBC is pursuing litigation against Atlas Roofing Corporation on behalf of people who purchased the company’s defective Chalet roofing shingles.
News
In a new safety alert, the FDA is warning of a dangerous risk of leg and foot amputations stemming from canagliflozin, the active ingredient in both Invokamet and Invokana, two widely used diabetes drugs. The FDA released the safety alert after reviewing the final results from two different clinical trials that looked at the drugs and their long-lasting effects over a large sample of patients suffering from type 2 diabetes.
If you have breast implants and were subsequently diagnosed with blood cancer, we are currently evaluating cases and would like to hear from you.
The FDA has recalled all Zimmer Biomet Comprehensive Reverse Shoulder replacement devices because of increasingly dangerous fracture rates. The FDA issued a Class I recall, saying that the Zimmer Biomet shoulder devices are fracturing at a higher rate than claimed by the manufacturer. The fracture rate raises the risk of serious injuries for patients who were implanted with the shoulder replacement device.
We are continuing to investigate claims for consumers throughout the U.S. who have been exposed to the weed killer Roundup. If you or a loved one have been diagnosed with non-Hodgkin lymphoma after exposure to Roundup, and would like more information about your rights, please contact us today.
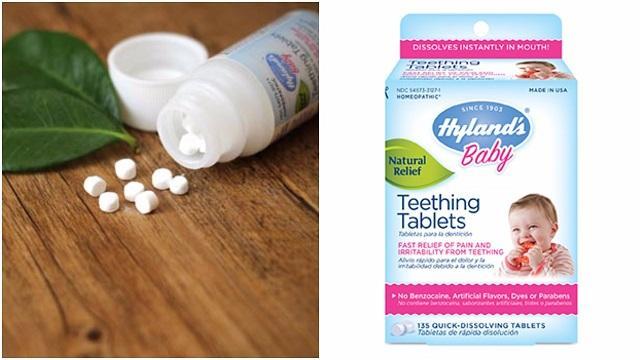
The team at Pendley, Baudin & Coffin is currently investigating cases involving injury to infants and children from Hyland’s homeopathic teething tablets and gels.
PBC is investigating and commencing litigation of cases against General Motors (GM) over claims involving its Generation IV 5.3 Liter V8 Vortec 5300 engine that was installed in 11 different SUV and truck models from 2010-2013. The claims say the defects cause internal damage to the engine.
For more information on the affected models, see our current case page.
Pendley, Baudin & Coffin, L.L.P. is proud to announce Chris Coffin's appointment to a top leadership position on the Plaintiffs' Steering Committee in the Taxotere multi-district litigation. As Co-Lead Counsel in the national litigation, Chris will be responsible for managing the Plaintiffs' Steering Committee which has been tasked with coordinating the efforts of all Taxotere permanent hair loss cases for plaintiffs across the U.S.
The attorneys at Pendley, Baudin & Coffin, L.L.P. are currently investigating potential legal action on behalf of patients who have been implanted with the Attune© Knee System made by DePuy Synthes, and are now having DePuy knee replacement implant problems.
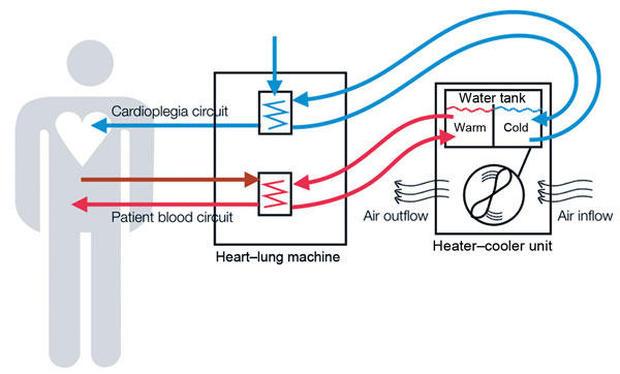
While modern medicine increasingly uses automated machines and devices to treat a wide variety of illnesses, the equipment itself can sometimes be linked to dangerous or even fatal issues. The public relies on federal regulators to adequately monitor these types of issues and ultimately ensure patient safety, but when that oversight fails, the consequences can be deadly.